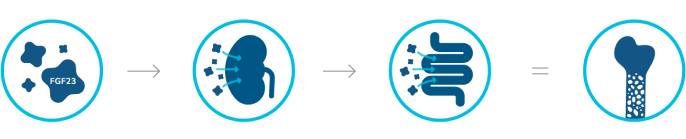
Excess FGF23
FGF23 is a protein hormone produced by osteocytes in the bones to regulate serum phosphorus levels. PHEX gene variants cause excess FGF23 activity.3
Reduced renal phosphorus reabsorption
Excess FGF23 results in excess phosphorus excretion.3
Decreased intestinal phosphorus absorption
Reduced active vitamin D results in decreased phosphorus absorption in the small intestine.3
Chronic hypophosphatemia
Due to loss of phosphorus, bones become weakened.2