Safety in children

CRYSVITA clinical safety profile in children with XLH1
Most common adverse reactions (≥10%) in CRYSVITA-treated patients observed in Study 1*
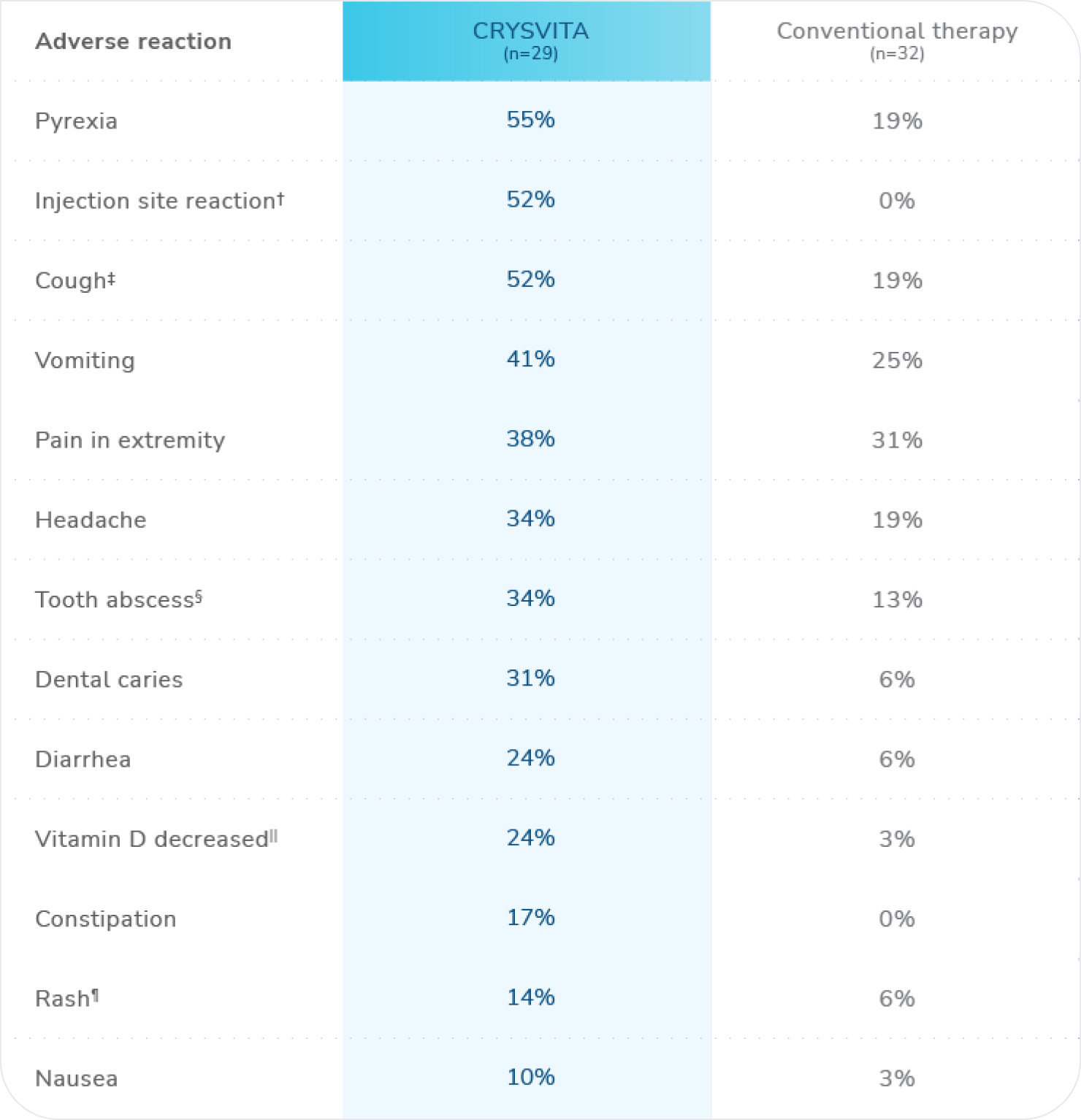
n=total number of patients who received at least 1 dose of CRYSVITA or conventional therapy.
* ≥10% in the CRYSVITA group that also occurred at a higher frequency than the conventional therapy group.1
† Injection site reaction includes: injection site reaction, injection site erythema, injection site pruritus, injection site swelling, injection site pain, injection site rash, injection site bruising, injection site discoloration, injection site discomfort, injection site hematoma, injection site hemorrhage, injection site induration, injection site macule, and injection site urticaria.1
‡ Cough includes: cough and productive cough.1
§ Tooth abscess includes: tooth abscess, tooth infection, and toothache.1
|| Vitamin D decreased includes: vitamin D deficiency, blood 25-hydroxycholecalciferol decreased, and vitamin D decreased.1
¶ Rash includes: rash, rash pruritic, rash maculopapular, rash erythematous, rash generalized, and rash pustular.1

Hypersensitivity reactions
The most frequent hypersensitivity reactions in the CRYSVITA arm were rash (10%), injection site rash (10%), and injection site urticaria (7%).1

Hyperphosphatemia
In pediatric studies, there were no events of hyperphosphatemia reported.1

Injection site reaction
Fifty-two percent of patients in the CRYSVITA arm had a local injection site reaction (eg, injection site urticaria, erythema, rash, swelling, bruising, pain, pruritus, and hematoma) at the site of CRYSVITA injection. Injection site reactions were generally mild in severity, occurred within 1 day of injection, lasted approximately 1 to 3 days, required no treatment, and resolved in almost all instances.1
Postmarketing experience
The following adverse reactions have been identified during postapproval use of CRYSVITA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood phosphorus increased in pediatric XLH patients only. Hypercalcemia, urticaria, and blood parathyroid hormone increased.

Ready to start your patients on CRYSVITA?
Take the first step by filling out the enrollment form.


Stay connected
Set up time with a representative to talk more about CRYSVITA,
or sign up for more information on CRYSVITA for the treatment of XLH.
